Description
Properties3D Printing
Mechanical properties
Thermal properties
Biological
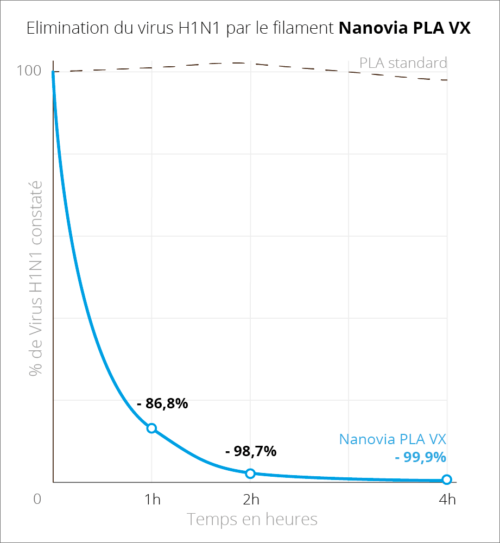
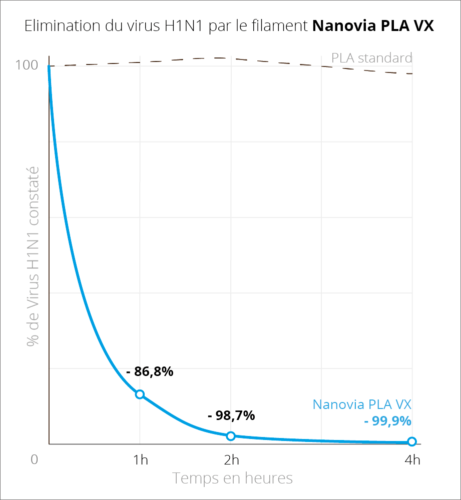
DocumentationDownload the technical chart Download the security chartlast updated : 17/05/2023 Print daily use objects with ease using Nanovia PLA VX, specially designed with additive fabrication in mind. This filament limits the spread and risks of biological contaminants, and is certified ISO 21702:2019. Nanovia PLA VX’s virucidal properties allow the elimination of 86.8% and 98.7% of the H1N1 virus, respectively after a contact period of 1 and 2 hours. Thanks to its radical action and safe for skin contact, Nanovia PLA VX is perfectly suited for the fabrication of everyday usage items. Virucide properties
The virucide test has been conducted according to the ISO 21702 norm by an independent laboratory in normalized conditions, on a FFF 3D printed test samples using H1H1 Influenza virus. Advantages
Other formatsThe Nanovia VX product range is composed of 2 types of 3D filaments, developed to limit the spread of infectious viruses :

Nanovia PLA VX is also available in pellet form for plastic extrusion and FGF 3D printing. User photosApplication recommendationsStorage
Health and safetyPost treatment
Use biocide products with precaution Please refer to the safety sheet and the technical data sheet when using Nanovia PLA VX ANSES N° : 20-07988 CertificationsPackagingVacuum packed spools, with desicant, packed in individual boxes with engraved serial number. Other formats available on demand.
FAQ:Is Nanovia VX certified efficiant avainst the Covid-19 Virus?Up to today there is no existing ISO validated norm proving the action against the Covid-19 virus. However, the Covid-19 is structured (corona virus) in the same family as the H1N1 virus. Is its protection longlasting ?Yes, the active charge of Nanovia VX is present throughout all of the material, allowing the surface to maintain its virucidal properties. It is important to make sure that the surface does not get covered to ensure its efficiency. Can printed parts be covered by another product (paints, varnish, etc…) ?No, it’s imperative to keep the surface of the printed parts such as, they cannot be covered with paint or varnish. In order to be efficient the material need to be in direct contact with the pathogen. Is it really efficiant?The biological test has been performed within the ISO 21702 norm by a certified independent laboratory on a 3D FFF printed test sample. Nanovia VX virucidal properties conform to the European norms. How do Nanovia VX virucidal properties work ?The biocidal active, with a complex copper and silver base, has been specially formulated to be incorporated in the Nanovia VX range. Our patented formulation allows for the presence of the decontamination agent throughout the printed parts and its stability over time. The active virucidal agent causes a degradation of the virus’ wall by catalysis. |